뉴스
GENIXCURE's treatment for dementia has been patented in the U.S
2021-01-11
GENIXCURE, a developer of a treatment for
degenerative brain diseases, announced on the 29th that it has obtained a U.S.
patent for a candidate substance for Alzheimer's dementia treatment.
ANL-101, which was patented this time, is a
treatment for Alzheimer's dementia, a degenerative brain disease. It is a new
drug candidate for gene therapy that completes treatment once administered, and
as a result of administering ANL-101 to Alzheimer's experimental model mice, it
has shown therapeutic effects such as significant improvement in cognitive and
learning ability.
ANL-101 is known as a gene therapy
substance that inhibits or promotes protein expression in the human body by
controlling "miRNA," which is a key key to aging.
The company said, "As a result of
administering ANL-101 to Alzheimer's model mice, the protein (E***) involved in
synaptic formation increased by 70% by controlling various target proteins at
once."
In addition, the company claims that the
number of proteins (S***) involved in cell protection and energy metabolism,
such as anti-aging, anti-inflammatory, and antioxidant, has increased by 60%,
which has been confirmed to activate nerve cell protection functions due to the
reduction of amyloid beta protein and inflammation.
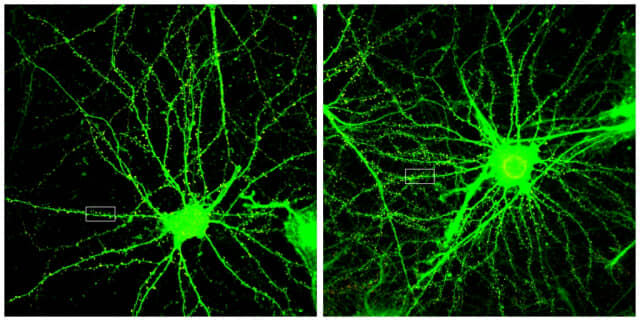
In addition, in mice administered with
ANL-101, the number of dendritic protrusions, a tentacle for receiving
information from nerve cells, increased by about 20%. This means that the
number of synapses in which signals are transmitted between nerve cells has
increased. An increase in the number of synapses means that nerve cell function
can be improved.
GENIXCURE plans to expand its indications to
mild cognitive impairment (MCI) and severe forgetfulness based on the high
cognitive function improvement effect of ANL-101 and the short- and long-term
memory and learning function improvement effect.
An official from GENIXCURE said,
"Dementia is the result of a number of complex factors acting at the same
time as various functions are degraded due to aging," adding, "We
proceeded with the development of new drugs with the confidence that cognitive
functions can be restored to normal levels only through complex
prescriptions."
Meanwhile, Kim Keetae, CEO of GENIXCURE, started this study in collaboration with the Institute of Basic Science (IBS) while working at the Daegu Gyeongbuk Institute of Science and Technology since 2012. After that, he applied for a patent in 2015. With the recent completion of U.S. patent registration, the company is pushing for full-scale commercialization of "degenerative brain disease treatment."

